
James Prescott Joule, the man
James Prescott Joule (24 Dec 1818-11 Oct 1889) was born in New Bailey St, Salford, Lancashire, and he was Baptised at Grosvenor St (Congregational) Chapel, Manchester, on the 9 Feb 1819.
His parents were Benjamin Joule and Alice Prescott who were married in Wigan, Lancashire, on the 14 May 1814. He was the second of five children and he was a member of a successful brewing family whose antecedents had
been Derbyshire farmers for many generations. James was a delicate boy due to a spinal ailment, which left him with a slight but permanent limp. Later in life, he was afflicted by frequent nose bleeding,
possibly caused by haemophilia.
James's elder brother was Benjamin St John Baptist Joule who was born on the 8 Nov 1817 in New Bailey St, Salford, Lancashire, and he was Baptised at Grosvenor Street (Congregational) Chapel, Manchester, on the 24 Dec 1817. Benjamin, besides being a landowner and Justice of the Peace, was a composer and possibly an author. In 1876 he owned 4,168 acres of land in County Donegal, Ulster. It is understood that he died on the 21 May 1895, aged 77 years, but his death was not recorded in England.
As children, private tutors taught James and Benjamin at home where James was particularly interested in scientific toys and he occupied himself among, other things, by flying kites in thunderstorms, in the manner of Benjamin Franklin. He also amused himself by giving family and servants electric shocks. One of James's tutors was the elderly John Dalton, 'the Father of Modern Chemistry', who had been reduced to earning money by teaching the children of wealthy families. Apparently, James's father was not impressed by John Dalton's tuition but it certainly made a lasting impression on James.
In 1847 he married Amelia Grimes, the daughter of the Comptroller of Customs at the Port of Liverpool. Tragically, she died in Salford in 1854 after only seven years of marriage. The couple had two sons and a daughter. Benjamin Arthur was born in 1850, Alice Amelia was born in 1852 and Henry James was born in 1854 but he died shortly afterwards. James was badly shaken by the loss of his wife and son and afterwards he became increasingly retiring. Although he was elected a Fellow of the Royal Society in 1850, he tended to remain remote from this. He also refused to take up a University post. During the last 17 years of his life he suffered from ill health and he died in Sale, Cheshire, on the 11 Oct 1889, aged 70 years.
James was buried in Brooklands Cemetery, Sale, Cheshire, his gravestone is inscribed with the number 772.55 and a quotation from the Gospel of St John, 'I must work the works of Him that sent me, while it is day: the night cometh, when no man can work' (9:4). Apparently, the number 772.55 was a value for the mechanical equivalent of heat that he obtained in 1878. There is also a statue of him in Manchester Town Hall.

James Prescott Joule.
James Prescott Joule, the physicist
When aged 15 years, Joule began work in his father's brewery where he took a keen interest in both engineering and chemistry. In those days, a brewery was the perfect place for him to study machinery, such as engines and
pumps that handled large quantities of liquids, and chemical processes, such as fermentation. Five years later he created a laboratory in the cellar of his parents' home and he began to attend the meetings of the
Manchester Literary and Philosophical Society, where he met famous scientists and engineers of the day. He flourished in the academic surroundings of the Society and he may never have formulated his principle of the
conservation of energy had he been educated at one of the country's universities because these were still governed by Newtonian mechanics.
Joule is best known for his determination of the mechanical equivalent of heat, for which he received honours, and, more recently, the honour of having his surname adopted as the Système International d'Unités (SI) unit of energy. The SI is the modern form of the metric system, finally agreed in 1960 by the supreme international authority on units of measurement, the General Conference of Weights and Measures (CGPM). Thus the SI supplementary unit of energy, work and quantity of heat is the joule, unit symbol J, this being the equivalent of one Newton metre (N m).
In Joule's day the imperial system of measurement of physical units was the foot-pound-second (FPS) system. This progressed through the centimetre-gram-second (CGS) system to the metre-kilogram-second (MKS) system before being replaced by the SI system that uses the three base units of the MKS system plus the ampere (electric current), mole (amount of substance), candela (luminous intensity) and kelvin (thermodynamic temperature).
To prove his theory that it was possible to convert mechanical work directly into heat Joule demonstrated in 1845, with the aid of a paddle-wheel that involved the paddle being driven in water by a falling weight suspended from a pulley, that the amount of work done always produced the same amount of heat. This enabled Joule to conclude that heat was a form of energy. After a number experiments he eventually concluded that 781.8 ft lb of work was needed to raise the temperature of one pound of water by one degree Fahrenheit. The order of magnitude of his results was considered by Joule to be compelling evidence of the convertibility of work into heat.
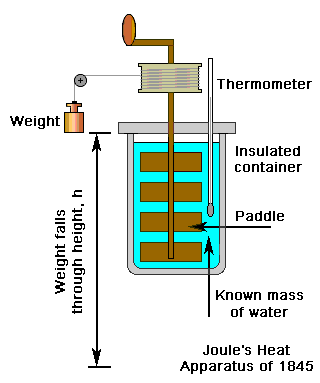
Hence Joule had determined the amount of mechanical work needed to produce an equivalent amount of heat, this quantity being known as 'the mechanical equivalent of heat'.
Nowadays, working in the original FPS system, it is known that the temperature of one pound of water is increased by one degree Fahrenheit when a weight of 777.65 lb falls from an altitude of one foot (or a weight of 1 lb falls through a height of 777.65 ft and so on).
In the modern SI system, the energy needed to raise the temperature of 1 kg of water by one degree kelvin is 4,184 J kg-1 K-1 and this is called the specific heat capacity of water.
By these experiments, Joule had discovered what became known as the First Law of Thermodynamics. This law of equivalence of heat and work may be stated as follows:
Heat and work are mutually convertible and in any action involving such conversion a definite quantity of heat is lost for every unit of work done and, conversely, a definite quantity of mechanical energy is used for every unit of heat gained. The relationship between work and heat is sometimes known as Joule's equivalent of heat.
Assuming 100% efficiency:
Heat absorbed by a material (J) = mass of material (kg) x specific heat capacity of the material (J kg-1 K-1) x temperature rise (K)
The First Law of Thermodynamics is the law of conservation of energy, where energy is the capacity to do work.
'Energy can neither be created or destroyed'
Another way of expressing the First Law of Thermodynamics is: The internal energy of a body (such as a fluid) can be increased by heating it and/or doing mechanical work on it.
In symbols: ΔE = Q + W, where E is the internal energy of the body, Q is the thermal energy, W is the work done (mechanical energy) and Δ is a finite increment in the variable E.
In 1840 Joule experimented on the power needed to drive electric motors and the heating effect of an electric current. He determined that the heat produced in a wire by an electric current is proportional to the square of the current multiplied by the resistance of the wire.
Hence: P = I2 R, where P is the power in watts, I is the current in amperes and R is the electrical resistance in ohms.
This relationship is known as Joule's Law.
Joule experimented with electric welding and also made the first mercury displacement vacuum pump. He proposed the constant pressure cycle for gas engines (Joule Cycle), which he said could run at much higher temperatures than steam engines and thus be more efficient. This cycle, now with the name 'Brayton Cycle', is used for gas turbines.
He also had an idea to change the power source of the family brewing business from steam power to electric power, the motors being supplied with electricity from batteries. Unfortunately, this idea was ahead of the technology of the day and the cost of zinc used in the manufacture of batteries was prohibitive. In his day it was still cheaper to produce steam for power by burning coal.
His papers on physics were apparently read to fellow members of the Manchester Literary and Philosophical Society at St Ann's Reading Room in Manchester, although the Society had purpose-built premises at 36 George St, which it occupied until the building was completely destroyed in the Christmas 1940 blitz.
In May 1847 he read his paper, 'On Matter, Living Force and Heat'. In this lecture he was able to give a more accurate figure for the mechanical equivalent of heat and this was 781.8 ft lb to raise the temperature of water by one degree Fahrenheit. In 1849 he read his memoir, 'On the Mechanical equivalent of Heat'. In this he stated, 'In accordance with the pledge I gave to the Royal Society some years ago, I have now the honour to present it with the results of the experiments I have made in order to determine the mechanical equivalent of heat with exactness.'
In 1882, Lord Kelvin said of him, 'His boldness in making such large conclusions from such small observational effects is almost as noteworthy and admirable as his skill in extorting accuracy from them.'
An amusing incident in Joule's life was to conduct a scientific experiment while on honeymoon in the Alps. He had always wanted to show that when water falls through 778 feet that its temperature rises by one degree Fahrenheit. His chance to do try this experiment came when the couple visited the falls at Chamonix. Armed with a large thermometer, he attempted to measure the rise in temperature at the bottom of the falls but he was thwarted in his attempt. The water did not fall through 778 feet and too much spray was produced to allow his experiment to work.
Joule was elected a fellow of the Royal Society in 1850 and his public honours included degrees from Dublin, Oxford and Edinburgh. He was awarded the Royal Medal of the Royal Society in 1852. In 1860 he was elected President of the Manchester Literary and Philosophical Society. In 1872 he was President of the British Association.
Census Returns for 1851, 1871 & 1881
1851 Census
| Name | Relationship to Head of Household | Marital Status | Age | Profession or Occupation | Birth Place |
|---|---|---|---|---|---|
| Benjamin JOULE | Head | Widower | 67 | Ale & Porter Brewer | Manchester, Lancs |
| Benjamin St John Baptist JOULE | Son | Widower¹ | 34 | Ale & Porter Brewer | Salford, Lancs |
| Mary JOULE | Daughter | Unmarried | 28 | Swinton, Lancs | |
| John JOULE | Son | Unmarried | 17 | Brewer | Swinton, Lancs |
| Jane WALLIS | Visitor | Unmarried | 38 | Langley, Derbys | |
| Rebecca NORTON | Nurse | Widow | 54 | House Servant | Wroxham, Norfolk |
| Elizabeth RIGBY | Servant | Unmarried | 44 | Do Do | Standish, Lancs |
| Margaret PURDY | Servant | Unmarried | 27 | Do Do | Rhuddlan, Flintshire |
| Mary HUGHES | Servant | Unmarried | 22 | Do Do | Barrow, Cheshire |
| Lavinia Amelia ALDRIDGE | Servant | Unmarried | 19 | Do Do | Weedon, Northants |
1871 Census
| Name | Relationship to Head of Household | Marital Status | Age | Profession or Occupation | Birth Place |
|---|---|---|---|---|---|
| James Prescott JOULE | Head | Widower | 52 | Landowner, DCL Oxon. LLD Dublin, Physicist | Salford, Lancs |
| Benjamin St. John Baptist JOULE | Brother | Married² | 53 | Landowner, Justice of the Peace for the County and Manchester | Salford, Lancs, |
| Mary JOULE | Sister | Unmarried | 48 | Housekeeper (Lady), Landowner | Swinton, Lancs |
| Benjamin Arthur JOULE | Son | Unmarried | 20 | Student at Owens College | Salford, Lancs |
| Alice Amelia JOULE | Daughter | Unmarried | 18 | Scholar | Salford, Lancs |
| Ann HUGHES | Servant | Unmarried | 37 | Domestic Cook | Barrow, Cheshire |
| Elizabeth JONES | Servant | Unmarried | 37 | Domestic Serving Maid | Chester, Cheshire |
1881 Census
| Name | Relationship to Head of Household | Marital Status | Age | Profession or Occupation | Birth Place |
|---|---|---|---|---|---|
| James Prescott JOULE | Head | Widower | 62 | DCL LLD, Physicist | Salford, Lancs |
| Mary JOULE | Sister | Unmarried | 58 | Annuitant | Salford, Lancs |
| Benjamin Arthur JOULE | Son | Unmarried | 30 | Artist (Painter) | Salford, Lancs |
| Sarah COWARD | Servant | Unmarried | 26 | Domestic Cook | ###side, Westmorland |
| Mary Adshead HEWITT | Servant | Unmarried | 22 | Domestic Housemaid | Sale, Cheshire |
Further Reading
James Prescott Joule manuscripts 1839–1887, The John Rylands Library, Manchester.
Joule, James Prescott, The scientific papers of James Prescott Joule, London: Taylor and Francis, 1884.
Cardwell, Donald S L, James Joule: A Biography, Manchester University Press, 1989.
Reynolds, Osborne, Memoir of James Prescott Joule, Cambridge University Press, 1892.